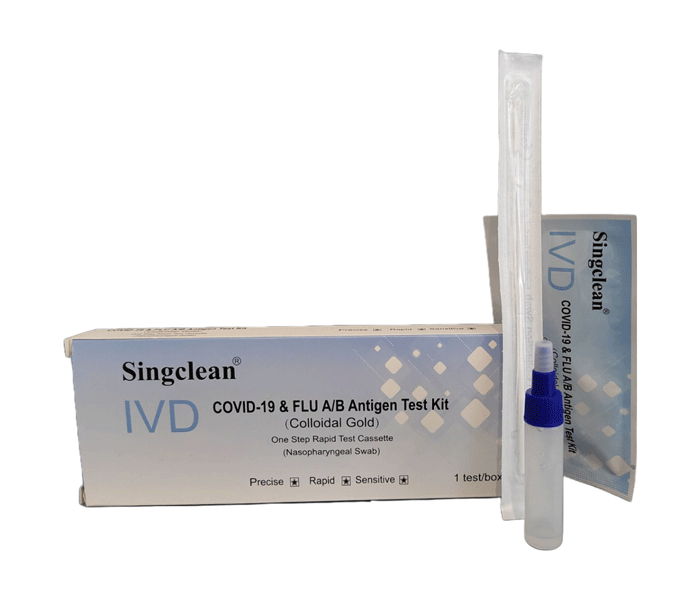
The IVD Covid-19 & FLU A/B Antigen (Colloidal Gold) is used for the in vitro qualitative detection of 2019 Novel Coronavirus antigen and Influenza A/B antigen in human nasopharyngeal swab specimens.
These two tests use colloidal gold immunochromatography to detect COVID-19 and Influenza A/B antigens.
The test for COVID-19 uses a monoclonal antibody to COVID-19 (T line of the test) and a polyclonal goat anti-mouse antibody (line C of the test) immobilized on a nitrocellulose strip.
The burgundy color conjugate patch contains colloidal gold conjugated to another monoclonal antibody to COVID-19. When the treated buffer containing the sample is added to the sample well, the COVID-19 will combine with the COVID-19 monoclonal antibody conjugate to form an antigen-antibody complex.
This complex migrates through the nitrocellulose membrane by capillary action. When the complex encounters the T-line COVID-19 monoclonal antibody line of the test, the complex is retained forming a burgundy colored band confirming a reactive test result. The absence of a colored zone in the test means a negative test result.
The Influenza A/B virus test is coated with Influenza A virus monoclonal antibody (Test lane A), Influenza B virus monoclonal antibody (Test B lane) and Goat anti-mouse polyclonal antibody (C lane of the test) on nitrocellulose membrane. The gold-labeled patch contains another influenza A virus monoclonal antibody and an influenza B virus monoclonal antibody conjugated to colloidal gold. When a sample containing Influenza A virus antigen is added to the sample well, the Influenza A virus antigen will combine with the Influenza A virus monoclonal antibody conjugate to form an antigen-antibody complex. The complex moves through the nitrocellulose membrane.
When the complex encounters the Influenza A virus monoclonal antibody in the A line of the test, the complex is retained and forms a burgundy zone, which confirms a positive test result. When a sample containing Influenza B antigen is added to the sample well, the Influenza B antigen will stain the B line of the chet, forming a burgundy line, thus confirming a positive Influenza B test result.
If there is no colored band on the A/B line of the test, the test result is negative.
These two tests contain a quality control line (test line C), which should show a purple-red color band of polyclonal goat anti-mouse antibody in combination with the colloidal gold conjugate on the gold label pad, regardless of the color in any other line of the test.
INTERPRETATION OF RESULTSCOVID-19 Antigen Detection Kit.NEGATIVE
The absence of any burgundy color in the T zone indicates that no COVID-19 antigen is detected in the sample. The result is negative.
positive
In addition to the presence of the C band, if the T band develops, the test indicates the presence of COVID-19 antigen in the sample. The result is positive for COVID-19.
error
The control bar does not appear. Insufficient sample volume or incorrect procedural techniques are the most likely reasons for failure to display a control line. Review the procedure and repeat the test with a new test cassette.
A/B Influenza Antigen Detection KitNEGATIVE
If only the C band is present, the absence of any burgundy color in the T band indicates that no Influenza A/B antigen is detected in the sample. The result is negative.
positive
Positive for Influenza A/B
In addition to the presence of the C line, if the A and B lines of the test appear at the same time, this means that both influenza A virus antigens and influenza B virus antigens are present in the sample, i.e. the result is positive for influenza A and influenza B.
Positive for Influenza A
In addition to the presence of the C line, if the A line of the test appears, the test indicates the presence of Influenza A antigen in the sample, i.e. the result is positive for Influenza A.
Positive for Influenza B
In addition to the presence of the C line, if the B line of the test appears, the test indicates the presence of Influenza B antigen in the sample, i.e. the result is positive for Influenza B.
cancel
The control bar does not appear. Insufficient sample volume or incorrect procedural techniques are the most likely reasons for failure to display a control line.
EAN: 6936020115056